- No products in the cart.
Automated DNA chemical synthesis
Over the last few decades, several approaches have been developed to synthesise short DNA sequences.
Relevant approaches include the phosphodiester method and the H-phosphonate method. However, higher yields and higher product quality and better efficiency and shorter synthetic times can be achieved using the solid-phase synthesis of oligonucleotides that employs nucleotide-3’-O-phosphoramidites as monomers.
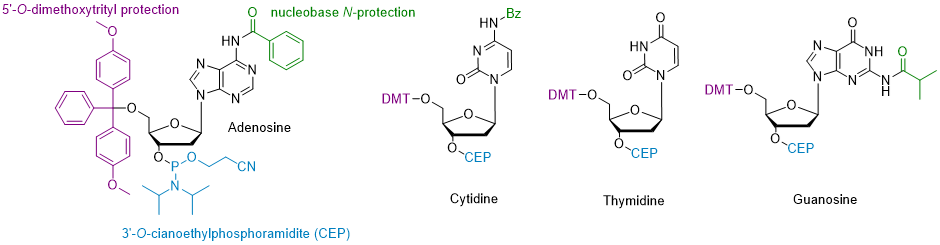
In this approach, the whole synthesis is conducted on solid support. Automated machines (called DNA synthesizers) have been developed to efficiently and quickly perform the synthesis. Building blocks for this synthesis are nucleotide-3’-O-cyanoethylphosphoramidites (CEP).
Generally, the 5’-hydroxyl group is protected with a dimethoxytrityl (DMT) group, while amino groups on nucleobases are protected with a benzoyl group (for adenosine and cytidine) or isobutyryl group (for guanosine).
Mechanism
At the beginning of the synthesis, the first nucleotide is attached to the solid support, loaded into a small column. The DNA synthesiser automatically carries on all the synthesis steps (shown in Scheme 1) by delivering necessary reagents and reactants to the column in which the oligonucleotide product is synthesised—the synthesis proceeds in the 3’ to 5’ direction (opposite to enzymatic synthesis). Finally, desired products are generally purified by standard desalting spin-columns or HPLC.
The main advantage of the solid-phase synthesis over traditional enzymatic synthetic approaches consists of a much larger possible scale. DNA oligonucleotides can be prepared up to μmols scale against the nmol scale of the enzymatic methods.
However, obtaining long DNA sequences via solid-phase synthesis is very difficult, and most reactive chemical groups have to be adequately protected.

Due to the vast and still quickly growing applications of non-natural oligonucleotides, the design and synthesis of non-natural building blocks for DNA synthesis is gaining outstanding importance.
If a non-natural nucleoside needs to be incorporated via solid-phase synthesis into the product sequence, its relative phosphoramidite has to be prepared and employed, where all other eventual reactive groups must be adequately protected. Also, acyclic or non-nucleoside phosphoramidites can be employed to insert a spacer within the product sequence or insert a modification at one end of the oligonucleotide.
@SantiagoLab, we can offer the design and synthesis of building blocks suitable for inserting a wide range of DNA modifications for different applications into your oligonucleotide products. If you would like to know more, or we can help you with your research, write an email to Krystof Sigut on krystof.sigut@santiago-lab.com or reach him on the phone +420 776 750 591.

