- No products in the cart.
CAS 1191237-80-5
€400 – €800
1191237-80-5 is provided by Santiago Lab (Prague, Czech Republic).
Purity (LC-MS)
99 %
Package contents
1191237-80-5
This compound is for research use only. We do not sell to patients.
| Size | Availability | Price | Quantity |
|---|---|---|---|
| 100 mg | In stock |
€400 | |
| 500 mg | In stock |
€600 | |
| 1 g | In stock |
€800 |
Characterisation
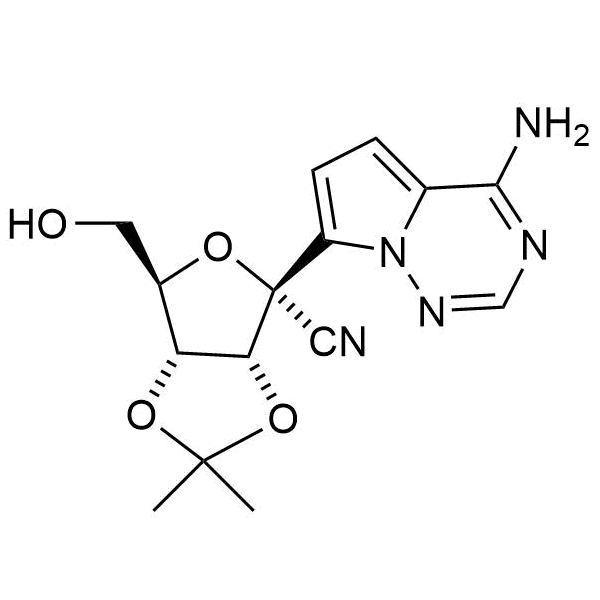
CAS: 1191237-80-5
IUPAC Name: D-Altrononitrile, 2-C-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-2,5-anhydro-3,4-O-(1-methylethylidene)-
Molecular weight: 331.33 g/mol
Molecular formula: C15H17N5O4
Description
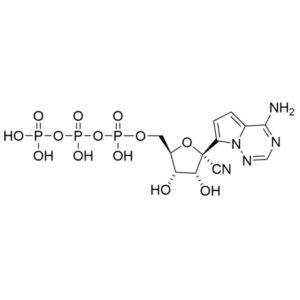
Nucleoside is an essential building block to the synthesis of Remdesivir triphosphate (1355149-45-9) and Remdesivir (1809249-37-3). Nucleoside derivate is used in the treatment of Feline infectious peritonitis (FIP). This fatal disease affects cats and is caused by a virus from a group of coronavirus. Remdesivir is nucleoside prodrug that metabolizes into GS-441524 which is phosphorylated to Remdesivir triphosphate. This phosphorus analog is mainly an active compound in the treatment of Ebola and COVID-19.
Chemicals are distributed worldwide
Buy 1191237-80-5 now, get your order in 48 hours
- Shipping through DHL in 48 hours
- All compounds are safely and rigorously packed
Payment
- We are sending the invoice the same day as the shipment
- We are able to modify the invoice for the academic institution, so the order can be paid from grants
| References | 1. Butler, T.; Cho, A.; Kim, C. U.; Saunders, O. L.; Zhang, L. Preparation of 1'-substituted carba-nucleoside analogs as antiviral agents. 2009-US41447, 2009132135, 20090422., 2009. |
|---|
Related products:
-
Aviromix
€700 -
Azithromycin – CAS 83905-01-5
€100 – €200 -
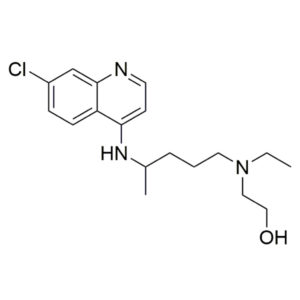
Hydroxychloroquine – CAS 118-42-3
€250 – €400 -
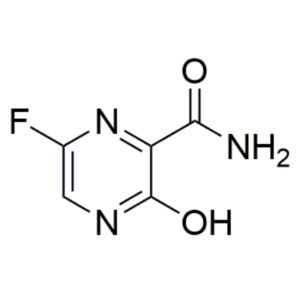
Favipiravir – CAS 259793-96-9
€200 – €350 -
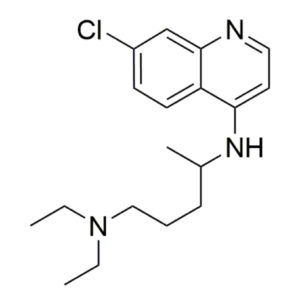
Chloroquine – CAS 54-05-7
€100 – €150