- No products in the cart.
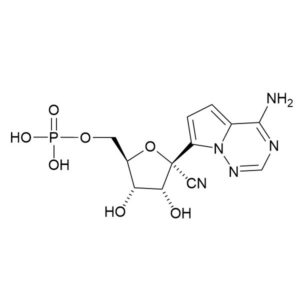
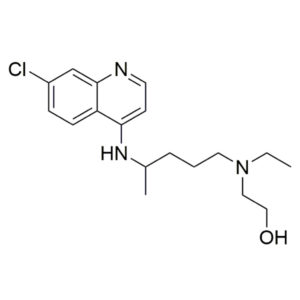
GS 441524 – CAS 1191237-69-0
Price range: €100 through €600
We cannot sell this compound for medical or veterinary use.
GS 441524 is provided by Santiago Lab (Prague, Czech Republic).
Purity (LC-MS)
99 % | Certificate of Analysis
Package contents
GS 441524
This compound is for research use only. We do not sell to patients or for veterinary use.
| Size | Availability | Price | Quantity |
|---|---|---|---|
| 10 mg | In stock |
€100 | |
| 50 mg | In stock |
€180 | |
| 100 mg | In stock |
€300 | |
| 500 mg | In stock |
€400 | |
| 1 g | In stock |
€600 |
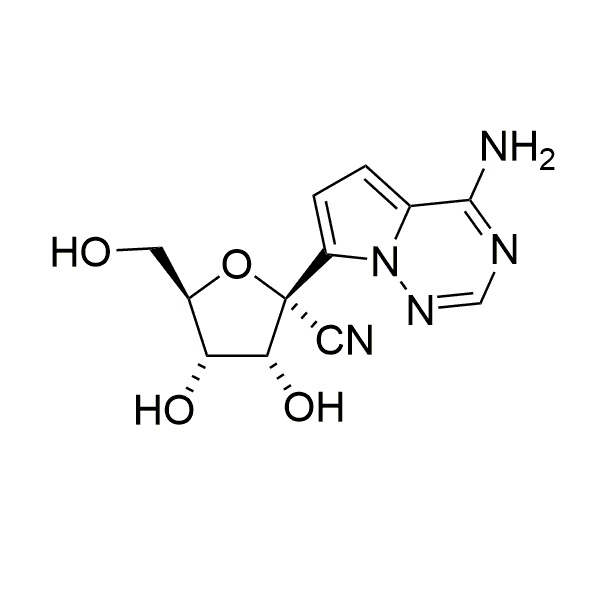
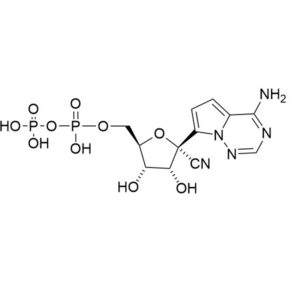
GS 441524
Characterisation
CAS: 1191237-69-0
IUPAC Name:(2R,3R,4S,5R)-2-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-carbonitrile
Other names: Nucleoside of Remdesivir; GS 441524
Molecular weight: 291.27 g/mol
Molecular formula: C12H13N5O4
Description
This nucleoside is an essential building block to the synthesis of Remdesivir triphosphate (1355149-45-9) and Remdesivir (1809249-37-3). Nucleoside derivate is used in the treatment of Feline infectious peritonitis (FIP). This fatal disease affects cats and is caused by a virus from a group of coronavirus. Remdesivir is a nucleoside prodrug that metabolizes into GS-441524 which is phosphorylated to Remdesivir triphosphate. This phosphorus analogue is mainly an active compound in the treatment of Ebola and COVID-19.
We are also offering Remdesivir and Remdesivir triphosphate.
We cannot sell this compound for medical or veterinary use.
Chemicals are distributed worldwide
Buy GS 441524 now, get your order in 48 hours
- Shipping through DHL in 48 hours
- Shipping worldwide for free.
- All compounds are safely and rigorously packed
Payment
- We are sending the invoice the same day as the shipment
- We are able to modify the invoice for the academic institution, so the order can be paid from grants
| References | 1. Butler, T.; Cho, A.; Kim, C. U.; Saunders, O. L.; Zhang, L. Preparation of 1'-substituted carba-nucleoside analogs as antiviral agents. 2009-US41447, 2009132135, 20090422., 2009. |
|---|
Related products:
-
Azithromycin – CAS 83905-01-5
Price range: €100 through €200 -
Favipiravir – CAS 259793-96-9
Price range: €200 through €350 -
Chloroquine – CAS 54-05-7
Price range: €100 through €150 -
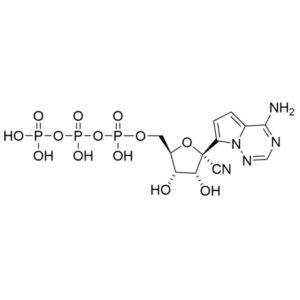
Remdesivir triphosphate – GS-443902 – CAS 1355149-45-9
Price range: €650 through €5090 -
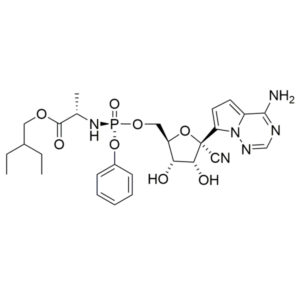
Remdesivir – CAS 1809249-37-3
Price range: €200 through €5900 -
Aviromix
€700 -
Remdesivir monophosphate – CAS 1911578-74-9
Price range: €1000 through €1500 -
Hydroxychloroquine – CAS 118-42-3
Price range: €250 through €400 -
Remdesivir diphosphate – CAS 1911578-77-2
Price range: €2500 through €3500