- No products in the cart.
Azithromycin – CAS 83905-01-5
€100 – €200
Azithromycin is provided by Santiago Lab. It is one of the 5 compounds recommended by WHO for the investigation of the treatment of COVID-19. You can order Azithromycin individually or as a set of 5 antivirotics recommended by WHO.
Purity (LC-MS)
98 % | Certificate of Analysis
Package contents
Azithromycin hydrate
This compound is for research use only. We do not sell to patients.
| Size | Availability | Price | Quantity |
|---|---|---|---|
| 100 mg | In stock |
€100 | |
| 1 g | In stock |
€200 |
Characterisation
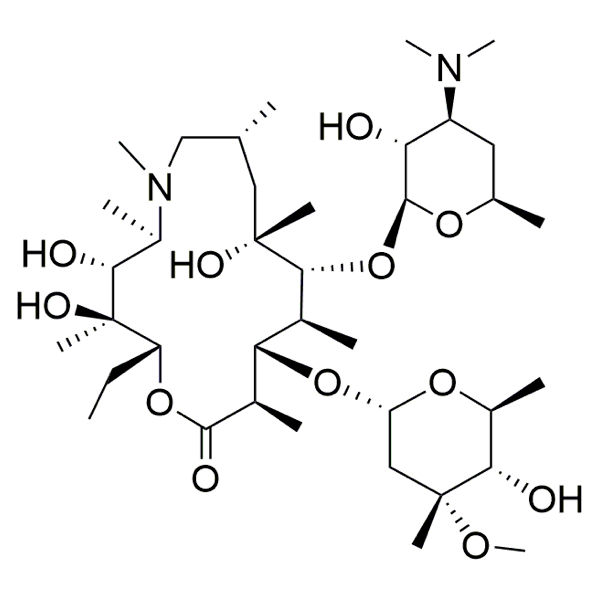
CAS: 83905-01-5
IUPAC Name: (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-11-[(2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy-2-ethyl-3,4,10-trihydroxy-13-[(2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyloxan-2-yl]oxy-3,5,6,8,10,12,14-heptamethyl-1-oxa-6-azacyclopentadecan-15-one
Other names: Azadose; Goxal; Sumamed; Ultreon; CP 62993; Zithromax; Azitrocin; Azythromycin; Azithromycin
Molecular weight: 749.00 g/mol
Molecular formula: C38H72N2O12
Description
Azithromycin is an antibiotic used for the treatment of a number of bacterial infections (middle ear infections, strep throat, pneumonia, traveller’s diarrhoea) or number of sexually transmitted infections (chlamydia and gonorrhoea infections). Along with other medications, it may also be used for malaria. Recently the combination of azithromycin with antiviral drug hydroxychloroquine was found to be more efficient in the treatment of COVID-19.
Azithromycin was discovered in 1980 by Pliva and approved for medical use in 1988. It is one of the Essential Medicines in the World Health Organization’s List, the safest and most effective medicines needed in a health system. Azithromycin belongs to family of azalides, a type of macrolide antibiotic. It works by decreasing the production of protein, thereby stopping bacterial growth.
Chemicals are distributed worldwide
Buy Azithromycin now, get your order in 48 hours
- Shipping through DHL in 48 hours
- All compounds are safely and rigorously packed
Payment
- We are sending the invoice the same day as the shipment
- We are able to modify the invoice for the academic institution, so the order can be paid from grants
| References | 1. Lover, A. A., Quantifying treatment effects of hydroxychloroquine and azithromycin for COVID-19: a secondary analysis of an open label non-randomized clinical trial (Gautret et al, 2020). medRxiv 2020. |
|---|
Related products:
-
Remdesivir – CAS 1809249-37-3
€200 – €5900 -
Chloroquine – CAS 54-05-7
€100 – €150 -
Favipiravir – CAS 259793-96-9
€200 – €350 -
Hydroxychloroquine – CAS 118-42-3
€250 – €400 -
Aviromix
€700