- No products in the cart.
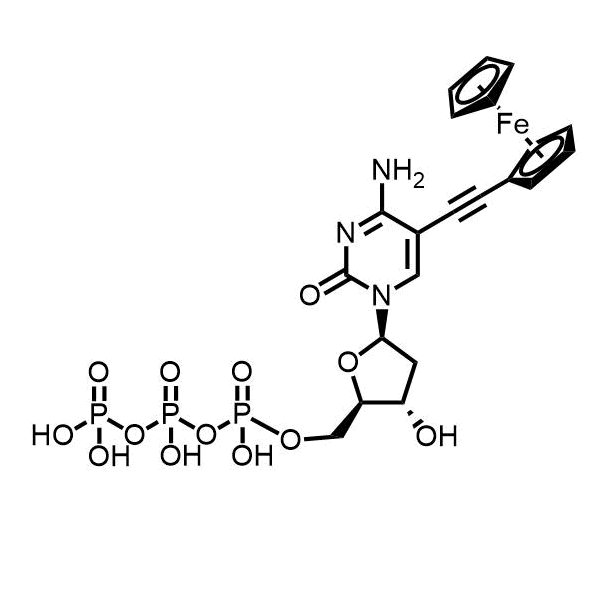
5-(Ferrocene-1-yl-ethynyl)-2′-deoxycytidine triphosphate – CAS 1150105-35-3
€1400 – €3500
5-(Ferrocene-1-yl-ethynyl)-2′-deoxycytidine triphosphate was synthesized and licensed at the IOCB Prague, lab of prof. Michal Hocek.
Purity (LC-MS)
99 %
Package contents
5-(Ferrocene-1-yl-ethynyl)-2′-deoxycytidine triphosphate Et3NH+ salt
This compound is for research use only. We do not sell to patients.
| Size | Availability | Price | Quantity |
|---|---|---|---|
| 1 mg | In stock |
€1400 | |
| 5 mg | In stock |
€3500 |
5-(Ferrocene-1-yl-ethynyl)-2′-deoxycytidine triphosphate – CAS 1150105-35-3
Characterisation
CAS: 1150105-35-3
IUPAC Name: 5-(Ferrocene-1-yl-ethynyl)-2′-deoxycytidine triphosphate
Molecular weight: 675.2 g/mol; 741.2 g/mol (Na+ salt)
Molecular formula:C21H26FeN3O13P3
Storage: -20 °C, protect from light
Description
5-(Ferrocene-1-yl-ethynyl)-2′-deoxycytidine triphosphate is a ferrocene labeled analog of natural cytidine triphosphate. Ferrocene is used as the gold standard for electrochemical labeling. Its conjugation at 5- position of cytidine triphosphate allows enzymatic incorporation of such analog in primer extension reactions and PCR. The compound might be used for electrochemical redox labeling of DNA, site-specific redox labeling, or in the development of highly sensitive genosensors.
5-(Ferrocene-1-yl-ethynyl)-2′-deoxycytidine triphosphate was synthesized and licensed at the IOCB Prague, lab of prof. Michal Hocek.
Chemicals are distributed worldwide
Buy 5-(Ferrocene-1-yl-ethynyl)-2′-deoxycytidine triphosphate now, get your order in 48 hours
- Shipping through DHL in 48 hours
- All compounds are safely and rigorously packed
Payment
- We are sending the invoice the same day as the shipment
- We are able to modify the invoice for the academic institution, so the order can be paid from grants
| References | 1. Brázdilová, P.; Vrábel, M.; Pohl, R.; Pivoňková, H.; Havran, L.; Hocek, M.; Fojta, M., Ferrocenylethynyl Derivatives of Nucleoside Triphosphates: Synthesis, Incorporation, Electrochemistry, and Bioanalytical Applications. Chemistry – A European Journal 2007, 13 (34), 9527-9533. |
|---|---|
| Gramáž | 1 mg, 5 mg |