- No products in the cart.
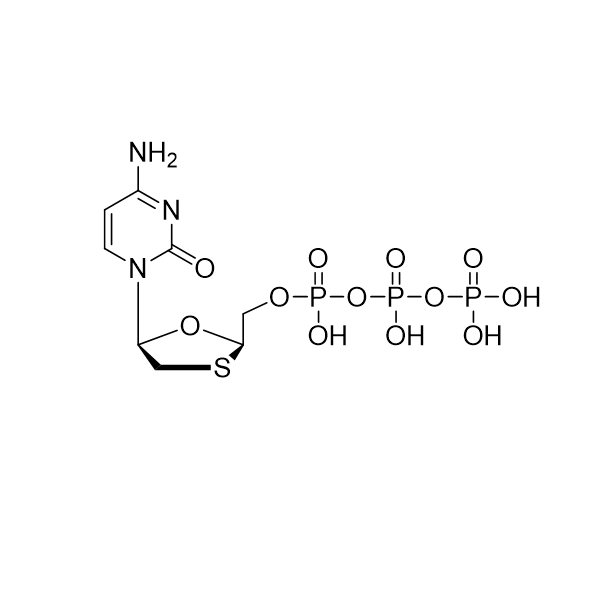
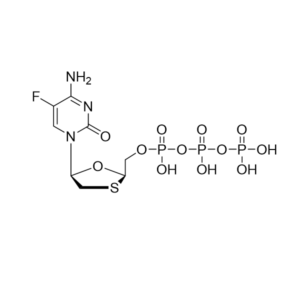
Lamivudine triphosphate – CAS 143188-53-8
€450 – €3500
Lamivudine triphosphate is synthesised by Santiago Lab (Prague, Czech Republic).
We are providing Lamivudine triphosphate in the form of Et3NH+ salt as a powder or water solution. All products are lyophilized and can easily sustain the shipment oversee.
Purity (LC-MS)
99%+ | Certificate of Analysis
Package contents
Lamivudine triphosphate Et3NH+ salt
This compound is for research use only. We do not sell to patients.
We are sending Lamivudine triphosphate in the form of Et3NH+ salt as a powder or water solution.
| Size | Availability | Price | Quantity |
|---|---|---|---|
| 1 mg | In stock |
€450 | |
| 5 mg | In stock |
€1900 | |
| 10 mg | In stock |
€3500 | |
| 100mM/10μL | In stock |
€450 | |
| 10mM/100μL | In stock |
€450 |
Lamivudine triphosphate
Characterisation
CAS: 143188-53-8, 2736455-56-2 (Et3NH+ salt)
IUPAC Name: Triphosphoric acid, P-[[(2R,5S)-5-(4-amino-2-oxo-1(2H)-pyrimidinyl)-1,3-oxathiolan-2-yl]methyl] ester
Other names: Lamivudine triphosphate, Lamivudine TP
Molecular weight: 469.20 g/mol,
Molecular formula: C8 H14 N3 O12 P3 S
Storage: -20 °C, protect from light, stored under argon
We are providing Lamivudine triphosphate in the form of Et3NH+ salt as a powder or water solution.
Description
Lamivudine triphosphate is synthesised by Santiago Lab (Prague, Czech Republic).
Lamivudine, commonly called 3TC, is an antiretroviral medication used to prevent and treat HIV/AIDS. It is also used to treat chronic hepatitis B when other options are not possible. It is effective against both HIV-1 and HIV-2. It is typically used in combination with other antiretrovirals such as zidovudine and abacavir. Lamivudine may be included as part of post-exposure prevention in those who have been potentially exposed to HIV. Lamivudine is taken by mouth as a liquid or tablet.
Common side effects include nausea, diarrhea, headaches, feeling tired, and cough. Serious side effects include liver disease, lactic acidosis, and worsening hepatitis B among those already infected. It is safe for people over three months of age and can be used during pregnancy. The medication can be taken with or without food. Lamivudine is a nucleoside reverse transcriptase inhibitor and works by blocking the HIV reverse transcriptase and hepatitis B virus polymerase.
Lamivudine was patented in 1995 and approved for use in the United States in 1995. It is on the World Health Organization’s List of Essential Medicines. It is available as a generic medication.
You can find more information about antiviral drug research at Santiago Lab in our article.
Chemicals are distributed worldwide
Buy Lamivudine triphosphate now, get your order in 48 hours
- Shipping through DHL in 48 hours. We can also arrange shipping on dry ice.
- All compounds are safely and rigorously packed
Payment
- We are sending the invoice the same day as the shipment
- We can modify the invoice for the academic institution so that the order can be paid from grants
| References | References |
|---|
Related products:
-
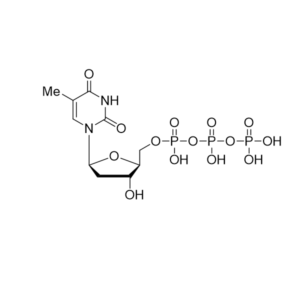
Telbivudine triphosphate – CAS 152502-95-9
€800 – €6500 -
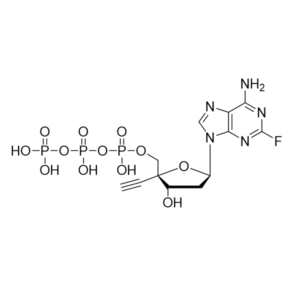
Islatravir triphosphate – CAS 950913-56-1
€2500 – €6000 -
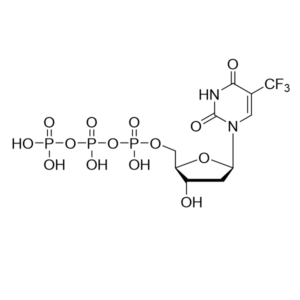
Trifluridine triphosphate – CAS 345-03-9
€800 – €6500 -
Emtricitabine triphosphate – CAS 145819-92-7
€700 – €2500 -
Clevudine triphosphate – CAS 174625-00-4
€800 – €6500