- No products in the cart.
Viruses are pathogens occurring all around us that can be fought with antiviral drugs. Viruses are the cause of a large number of diseases, many of which are deadly – from the common cold, influenza, herpes, to Ebola, AIDS, and recently also COVID-19.
Experts on viruses have not yet agreed on whether the viruses are alive or not. Viruses are a piece of genetic information wrapped in protein cover, and their main goal is to multiply. To do that, they need to get into the host cell and force it to replicate its genetic information and produce proteins essential for viral assembly.
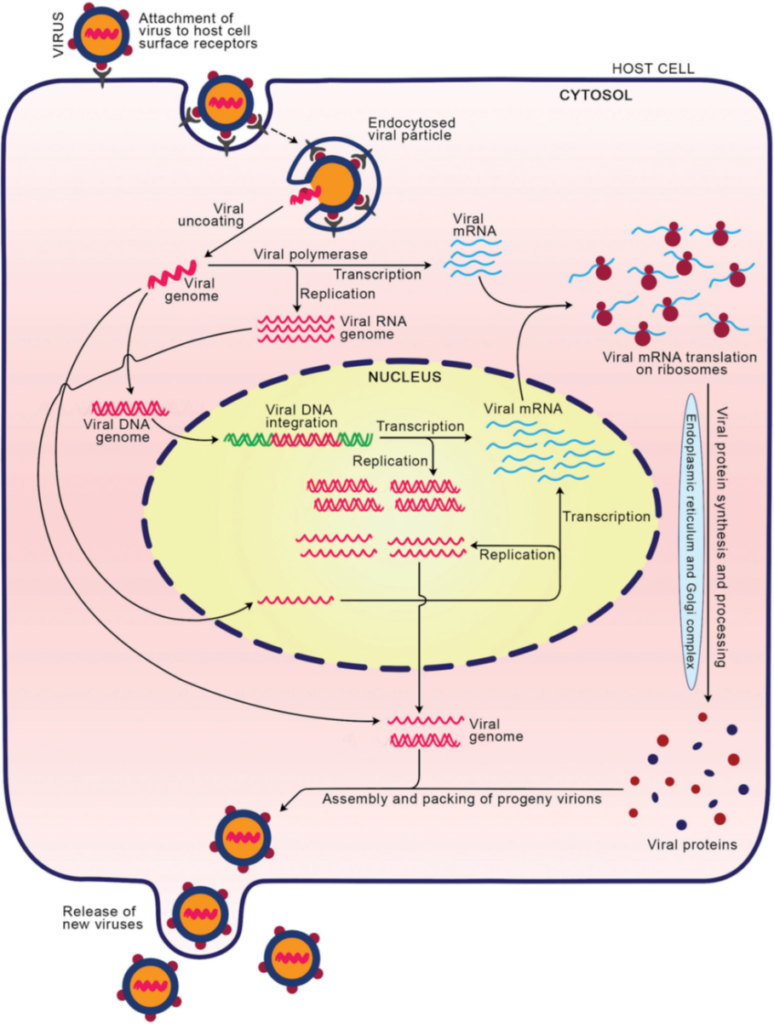
Viruses can be divided into several classes based on their genetic information and the mechanism of their reproduction.
The main classes are DNA viruses, RNA viruses and reverse-transcribing viruses.
DNA viruses possess either single-strain DNA or double-strain DNA. The examples of this class are Herpes simplex virus, hepatitis B virus, smallpox and chickenpox viruses, Epstein-Barr virus and some viruses causing the common cold.
RNA viruses have their genetic information stored in RNA and proceed replication using RNA-dependent-RNA-polymerase. It is the biggest group of viruses, including the ones causing diseases like common cold (rhinovirus), polio (poliovirus), Dengue fever and other haemorrhagic fevers (Ebola, Margburg), yellow fever, Zika, hepatitis C, flu (influenza viruses), rabies, measles, mumps and coronavirus-caused respiratory diseases (SARS, MERS, SARS-CoV-2).
Reverse transcribing viruses use reverse transcriptase to create DNA, then integrate into the host genome. The well-known example of this class is HIV (having an RNA genome), which causes AIDS.
Fighting the virus
There are several possibilities for how to fight viral infections. The first option is vaccination, as there are a lot of available vaccines against a large number of viruses. Some viral diseases have been practically eradicated because of extensive immunisation, such as measles, polio and smallpox.
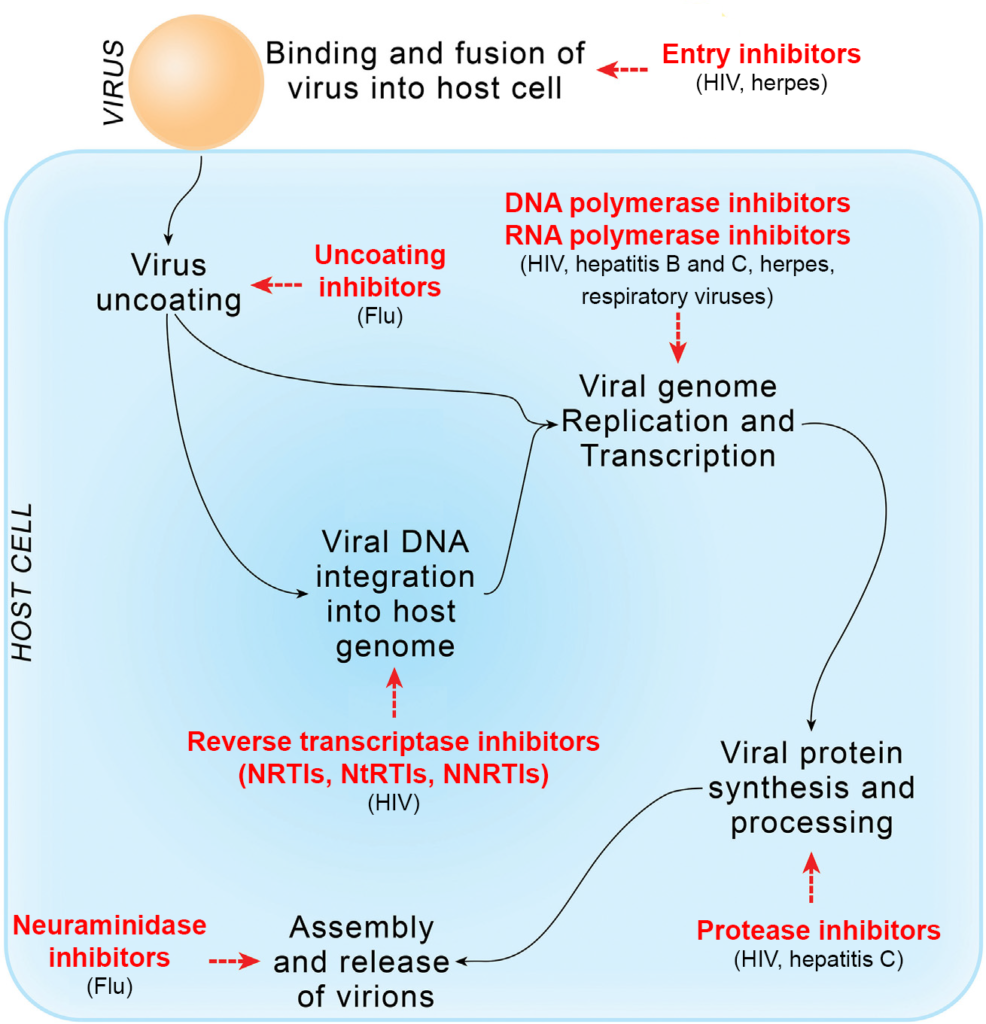
Another option is to target the viruses for drug treatment. The drugs developed to selectively affect viruses are called antiviral drugs or antivirotics. They are designed to disrupt key phases of the viral replication cycle, such as cell entry, uncoating of the viral particle, genome transcription, viral particle assembly or its release from the host cell. Most of the antiviral drugs currently available for treatment are those used to treat herpes, hepatitis B and C, influenza and HIV infection.
Entry inhibitors
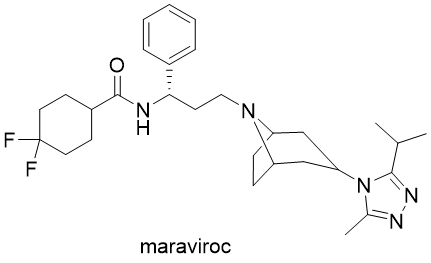
Compounds from this class are used mainly in the treatment of HIV infection. They interfere with binding, fusion and entry of virions (active or intact virus particle) to human cells. Entry inhibitors are often used in combination therapy with different antiviral drugs. Examples of approved drugs are small molecules maraviroc (brand names Selzentry (US) and Celsentri (EU)) and fostemsavir (Rukobia), together with oligopeptides enfuvitridine (Fuzeon) and bulevirtide (Hepcludex), which is used for the treatment of hepatitis B and D.
Antiviral drugs- uncoating inhibitors
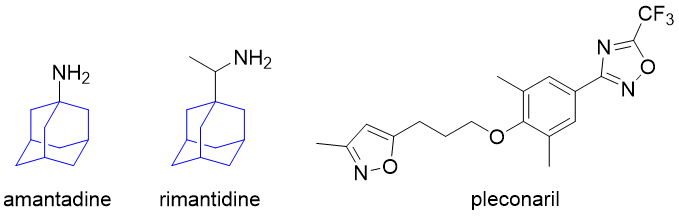
To make the viral genetic information available for replication, the virus’s protein coat (or envelope) must be disrupted or removed. This process is called uncoating. Adamantane derivatives amantadine and rimantadine are the two examples of uncoating inhibitors used in the treatment of influenza; however, their use is limited nowadays because of increasing viral resistance. Another example is pleconaril, which works against rhinoviruses (such as a common cold) or enteroviruses, which can cause diarrhoea, meningitis or encephalitis.
Antiviral drugs- inhibitors of the genome replication process
The main class of antiviral drugs consists of compounds acting as inhibitors of transcription processes, such as inhibitors of reverse transcriptase (retroviruses), inhibitors of DNA polymerase (DNA viruses) or inhibitors of RNA polymerase (RNA viruses). These compounds are mainly nucleoside analogues or non-nucleoside transcription inhibitors. In most cases act as prodrugs that are metabolised into the active form (such as nucleoside triphosphates) in the human body.
To treat retroviral infections such as HIV, both nucleoside/nucleotide and non-nucleoside reverse transcriptase inhibitors (NRTIs/NtRTIs and NNRTIs) are used.
Examples of nucleoside inhibitors are zidovudine (AZT, Retrovir), the first FDA approved antiretroviral drug against HIV, stavudine (Zerit – HIV), lamivudine (3TC, Zeffix, Epivir – HIV and hepatitis B), emtricitabine (Emtriva – HIV, clinical trials for hepatitis B), abacavir (ABC, Ziagen – HIV) and entecavir (ETV, Baraclude – hepatitis B).
Commonly used are phosphonate-based nucleotide analogues tenofovir and adefovir, developed by prof. Antonín Holý at the Czech Academy of Sciences together with Gilead Sciences. Prodrug of tenofovir, tenofovir disoproxil fumarate (TDF, Viread) is one of the most used anti-HIV drugs, adefovir (ADV, Preveon, Hepsera) is approved for the treatment of hepatitis B. The combination of emtricitabine and TDF is approved in the US with the trade name Truvada.

@Santiagolab, we have successfully prepared active triphosphates of lamivudine and emtricitabine, as well as triphosphates of other nucleoside derived drugs, and we offer them to researchers worldwide. Check our website for more information.
Non-nucleoside reverse transcriptase inhibitors are often used in combination with other antiretroviral drugs. Examples of approved compounds are efavirenz, nevirapine, delavirdine, etravirine, rilpivirine and doravirine (MK-1439, Pifeltro), which is also used in combination with lamivudine and TDF under the brand name Delstrigo.
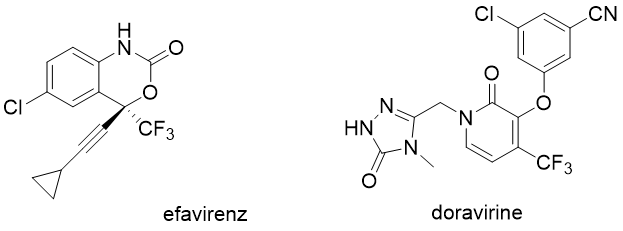
Recently, a novel nucleoside analogue islatravir has advanced into later phases of clinical evaluation. You can read more about its structure, unique properties, mechanism of action and potential in HIV treatment in a separate article previously published on our blog.
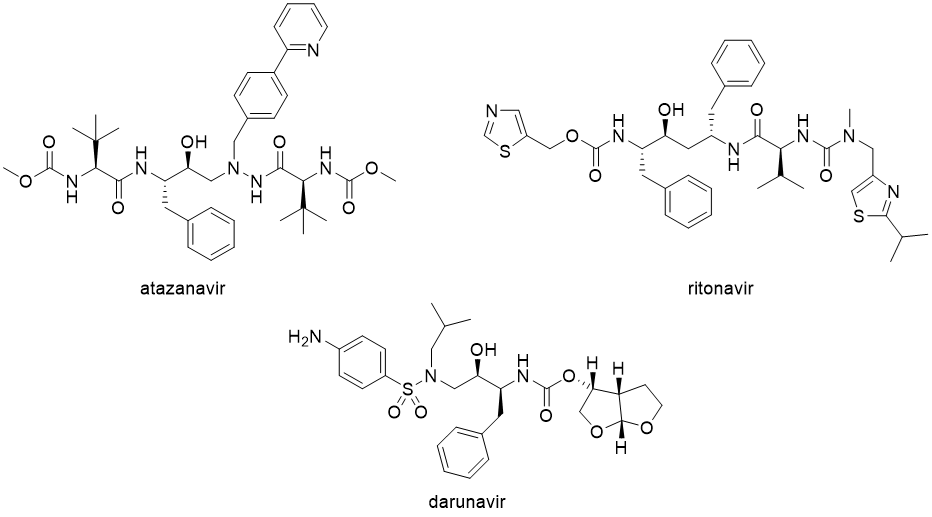
Another class of antiretroviral drugs is protease inhibitors, which interfere with the production of proteins essential for the assembly of the new virion particle. Approved drugs from this class are atazanavir (Reyataz), darunavir (Prezista), fosamprenavir (Lexiva), indinavir (Crixivan), lopinavir, ritonavir (Norvir), nelfinavir, saquinavir, tipranavir and cobicistat.
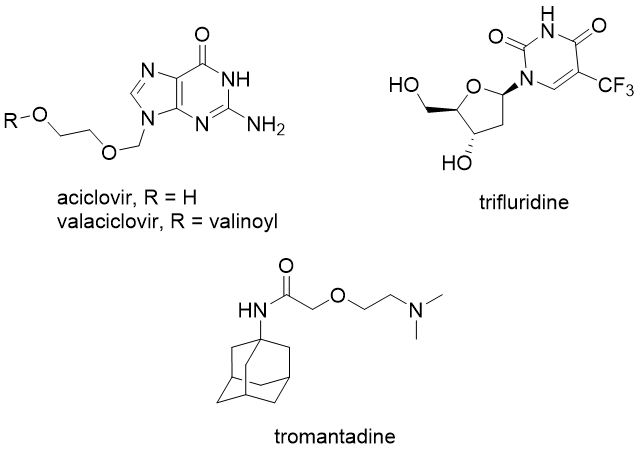
Herpes is a disease caused by the herpes simplex virus, belonging to the class of DNA viruses. There are several drugs for treating this disease, such as aciclovir, valaciclovir, edoxudine, idoxuridine, penciclovir, trifluridine, vidarabine. All of them inhibits viral DNA polymerase and thus prevent viral DNA replication. Other used antiviral compound against herpes is tromantadine, an adamantane derivative that acts as an entry and uncoating inhibitor.
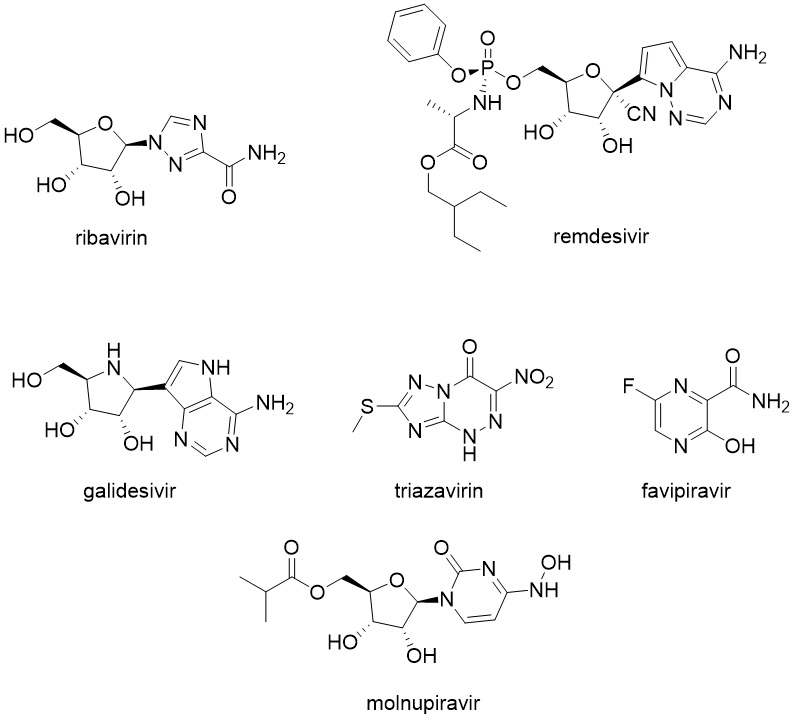
Only a few drugs are approved for everyday use for the broad class of RNA viruses. Some of them are targeted explicitly as RNA polymerase inhibitors, including sofosbuvir and ribavirin against hepatitis C and remdesivir, approved against covid-19.
Other drugs against RNA viruses can be categorised as broad-spectrum antiviral drugs, and most of them are experimental or approved only in a limited number of countries. These drugs are often nucleoside analogues, and they are transformed in the human body to their active form, primarily as triphosphate derivatives.
There are around 150 known broad-spectrum antiviral drugs in different stages of development with reported activity against almost 80 other viruses. These compounds often show activity against various viruses than investigated initially and could be promising in drug repurposing and as potential candidates for the treatment of re-emerging viruses such as Ebola, Marburg and SARS-CoV-2. Some examples of this class of compounds are favipiravir (approved in Japan, recommended in China against covid-19), galidesivir (originally developed against hepatitis C), triazavirin (developed in Russia) and molnupiravir, a promising drug for the treatment of covid-19 which was recently approved in the UK.
To get more information about molnupiravir, read our previously published article on our blog.
As mentioned above, broad-spectrum antiviral drugs have the potential for drug repurposing. Together with the emergence of covid-19 pandemics, we have prepared Aviromix, a set of potential anti-covid drugs, to help researchers worldwide search for possible treatment. Check out Aviromix on our website.
The last category of antiviral drugs is neuraminidase inhibitors, the enzyme essential for the release of influenza viral particles from host cells. These compounds are used in the treatment of influenza and include oseltamivir (Tamiflu), zanamivir (Relenza) and peramivir (Rapivab).
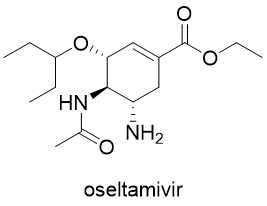
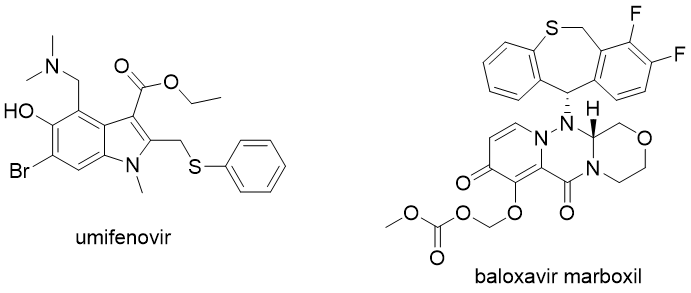
There are some other anti-influenza drugs with limited approval. Umifenovir (Arbidol) is a drug approved in Russia that acts as an entry inhibitor. Baloxavir marboxil is an anti-influenza drug approved in Japan and the USA with a different mode of action. It inhibits the influenza virus’ cap-dependent endonuclease activity, thus interfering with the production of viral mRNAs.
@SantiagoLab, we can offer the design and synthesis of various antiviral compounds as well as the preparation of triphosphates of desired nucleosides. If you would like to know more, or we can help you with your research, write an email to Krystof Sigut on krystof.sigut@santiago-lab.com or reach him on the phone +420 776 750 591.
Drop us a line
We will respond as soon as possible, typically within 48 hours.
References
Tompa, D.R.; Immanuel, A.; Srikanth, S. et al. Trends and strategies to combat viral infections: A review on FDA approved antiviral drugs. International Journal of Biological Macromolecules 2021, 172, 524–541.
Bekerman, E.; Einav,S. Combating emerging viral threats. Science 2015, 348 (6232), 282–283.
Baltimore, D. Expression of animal virus genomes. Bacteriol Rev. 1971, 35 (3), 235–241.


