- No products in the cart.
The discovery of the first antibiotic, penicillin (Figure 1), by Alexander Fleming in 1928 was a major breakthrough in the treatment of various, until that time fatal, infections.
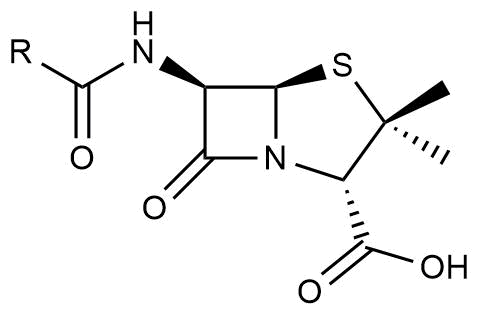
Figure 1. Penicillin core structure motive.
The 1960s and 1970s have witnessed a peak in optimism regarding control of bacterial infections. It was widely believed that the major classes of antibiotics, β-lactam, quinolone, tetracycline, and macrolide, would provide essential human protection against all bacterial pathogens.
However, the spread of methicillin-resistant Staphylococcus aureus (MRSA) in the 1980s and 1990s clearly showed that developing new substances with antibiotic effects is a necessity.
To this day, an enormous synthetic effort of organic chemists helped to discover and develop many new promising antibiotics. You can see the major classes of these antibiotics in the beautiful picture below (Figure 2, thanks to
https://www.compoundchem.com/2014/09/08/antibiotics/).
for creating it).
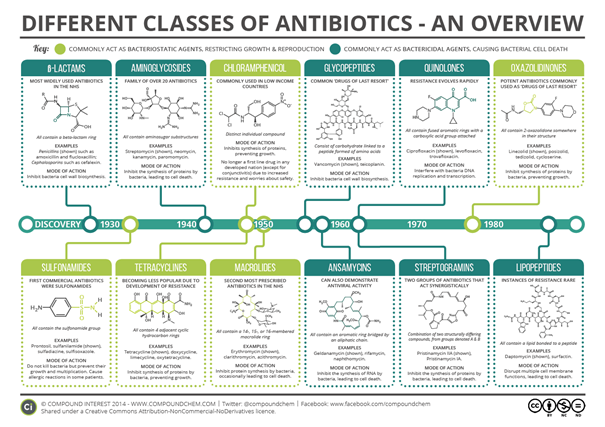
Figure 2. An overview of antibiotics classes from https://www.compoundchem.com/2014/09/08/antibiotics/).
In our lab, in collaboration with the Institute of Microbiology of the Czech Academy of Sciences (https://lab111.mbu.cas.cz/index.php/en/?jjj=1591019119554), we are studying another class of antibiotics, lincosamides. These compounds can prevent bacterial replication by blocking the synthesis of their proteins.1,2
So far, only naturally isolated lincomycin and semi-synthetic derivative clindamycin are commonly used in clinical practice (Figure 3). This is mostly due to the issues with low biological activity or high toxicity of other synthetically prepared lincosamides.
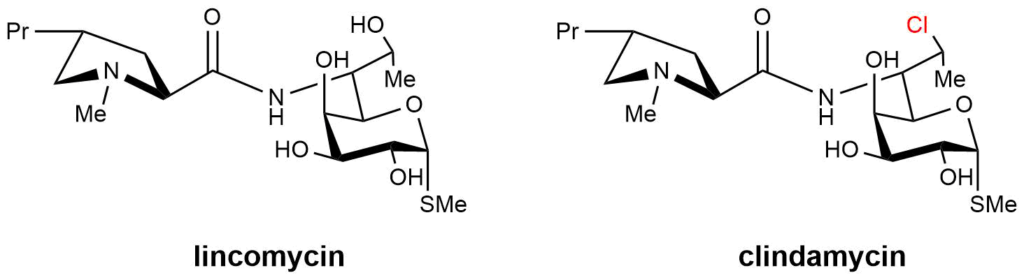
Figure 3. Structures of commonly used lincosamides.
We have worked on lincosamide antibiotic development over the past two years at Santiago Lab. We have obtained a significant amount of experience with their challenging synthesis, purification, and analysis.
During this time, we have encountered many complications both in the synthesis and analysis. For example, since these compounds have surprisingly low UV absorption, we had to develop reliable analytical techniques to follow the course of the synthesis.
Furthermore, it is not always easy to transform the synthesis from mg scale to gram scale. But about these challenges, we will write soon in another post dedicated to scale-up in our laboratories.
Recently, we have started working in this area even more profound, and we are now in the process of developing a library of structurally modified lincosamides (Figure 4).
Compared to the commercially available lincosamides, these compounds will offer better activity against various bacteria (such as Methicillin-resistant Staphylococcus aureus– MRSA or Streptococcus pneumoniae) compared to the commercially available lincosamides (see Figure 3).
Moreover, due to the synergic effects of structural motives in the molecule, these compounds will be much safer to use.
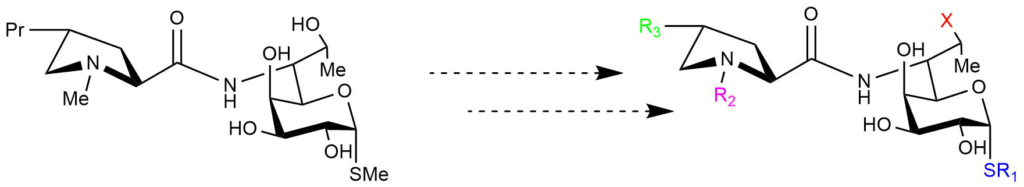
Figure 4. Development of new lincosamide antibiotics.
If you would like to know more about antibiotics, or if we can help you with your research, write an email to Krystof Sigut on krystof.sigut@santiago-lab.com or reach him on the phone +420 776 750 591.
Drop us a line
We will respond as soon as possible, typically within 48 hours.
References
- Kadlčík, S., Kameník, Z., Vašek, D., Nedvěd, M., Janata, J. (2017). Elucidation of salicylate attachment in celesticetin biosynthesis opens the door to create a library of more efficient hybrid lincosamide antibiotics. Chemical Science 8(5), 3349-3355.
- Janata, J., Kamenik, Z., Kadlcik, S., Najmanova, L., Gazak, R. (2019). Lincosamide derivatives, preparation and use thereof as antimicrobial agent. International patent application, WO2018161979.


