- No products in the cart.
AIDS is a terminal disease caused by the human immunodeficiency virus (HIV), affecting millions worldwide.
HIV belongs to the group of retroviruses, which act by incorporating the genetic information stored in the viral RNA into the host DNA using the reverse transcription. Therefore, retroviruses, in general, are very hard to be fought against and to this day, there is no definitive treatment of HIV.
AIDS was once a fatal disease, but since the introduction of highly active anti-retroviral therapy (HAART) in 1996, HIV infection has become a long-lasting disease. Although anti-HIV drugs must be administered on a regular basis for the rest of the life to suppress the viral activity and the symptoms, the introduction of HAART meant a huge improvement of the quality of life and survival rate of HIV infected patients.
One of the most currently used anti-HIV drug tenofovir (TFV), and its prodrug tenofovir disoproxil fumarate (TDF), were developed by the famous Czech scientist Antonín Holý together with Gilead Sciences.
Islatravir
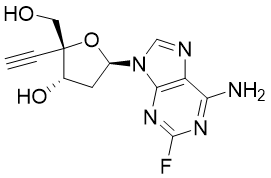
Islatravir (ISL, MK-8591, EFdA, CAS: 865363-93-5) is an investigational antiviral drug developed by Merck for the treatment of HIV. Currently, it is one of the hottest anti-retroviral agents, as it has been advanced to phase 2 or 3 in several clinical trials for HIV treatment.
There are several classes of anti-retroviral drugs – HIV protease inhibitors, entry (or fusion) inhibitors, and non-nucleoside or nucleoside/nucleotide reverse transcriptase inhibitors (NNRTIs and NRTIs). The most effective HIV treatment uses a combination of agents from at least two different classes, but there has been a problem lately with the emergence of drug-resistant HIV mutants.

Islatravir inhibits HIV reverse transcriptase, but has a different mechanism of action with respect to all other approved NRTIs because of its unique structural features: a 3’-hydroxyl group makes this nucleotide a translocation inhibitor rather than a chain terminator. The introduction of a 4’-ethynyl group enhances the selectivity towards the HIV reverse transcriptase. Similarly to other nucleoside antivirotics, islatravir is first transformed to islatarvir-5’-triphosphate (ISL-TP), which is the biologically active form of islatravir. When ITP is incorporated into forming DNA strand by the reverse transcriptase, it acts as a chain terminator by preventing the translocation.
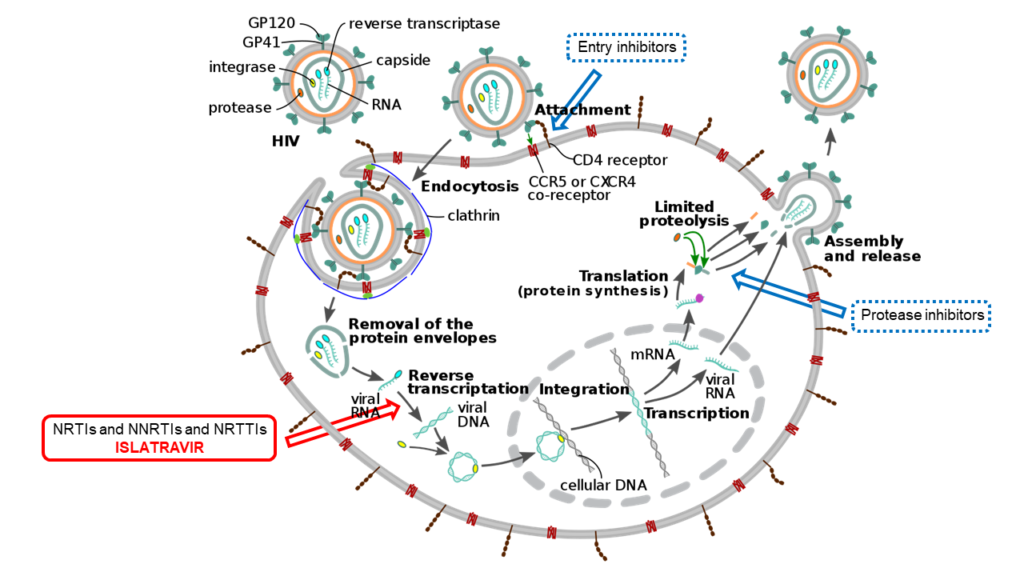
Replication cycle of HIV in CD4 white blood cell with possible targets for the treatment of infection
Islatravir has a higher anti-HIV activity than currently used NRTIs zidovudine (AZT), lamivudine (3TC), emtricitabine (FTC) and TDF, and also shows a substantial effect on multidrug-resistant HIV strains. Moreover, its active form ISL-TP has a long half-life in peripheral blood cells.
Based on its unique properties, Merck is currently developing a sub-dermal implant for the long-term release of Islatravir. This implant is now under clinical evaluation and is likely to cause a revolution in HIV treatment.
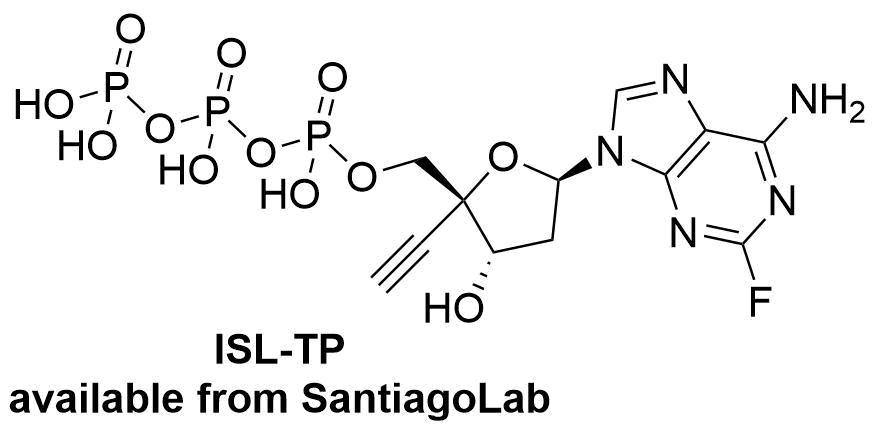
@SantiagoLab, we have successfully employed the synthesis of islatravir-5’-triphosphate (ISL-TP), the biologically active form of islatravir. Together with other nucleoside triphosphates, we offer ISL-ITP in excellent quality for research purposes to scientists worldwide.
If you would like to know more about Islatravir or Islatravir triphosphate, or if we can help you with your research, write an email to Krystof Sigut on krystof.sigut@santiago-lab.com or reach him on the phone +420 776 750 591.


