- No products in the cart.
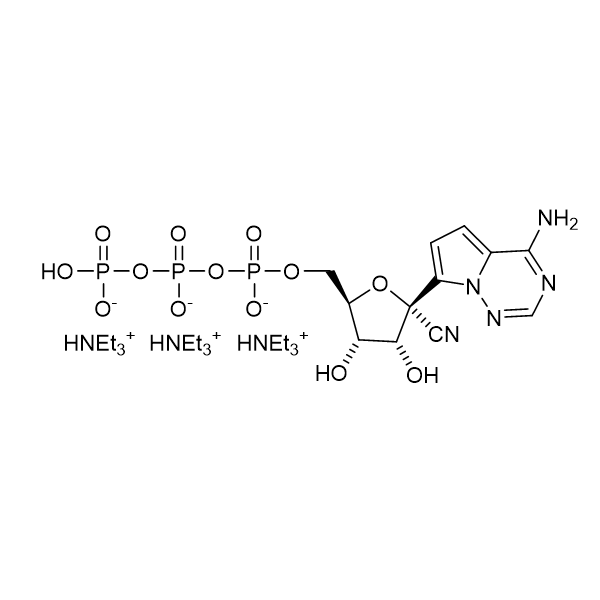
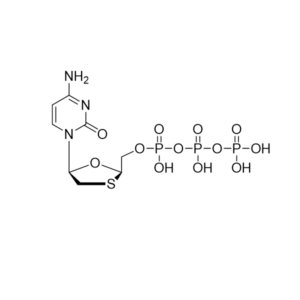
Remdesivir triphosphate triethylammonium – GS-443902 – CAS 2725956-16-9
Price range: €650 through €5090
Remdesivir triphosphate triethylammonium salt is synthesised by Santiago Lab (Prague, Czech Republic).
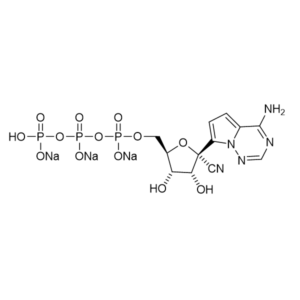
All products are lyophilized and can easily sustain the shipment oversee (shipping with DHL on dry ice to ensure that product arrives in a perfect state). We are also providing Remdesivir triphosphate in the form of Na+ salt.
Purity (LC-MS)
99%+ | Certificate of Analysis
Package contents
Remdesivir triphosphate triethylammonium salt
This compound is for research use only. We do not sell to patients.
| Size | Availability | Price | Quantity |
|---|---|---|---|
| 1 mg | In stock |
€650 | |
| 5 mg | In stock |
€780 | |
| 10 mg | In stock |
€1115 | |
| 25 mg | In stock |
€1960 | |
| 50 mg | In stock |
€3220 | |
| 100 mg | In stock |
€5090 | |
| 100mM/10μL | In stock |
€650 | |
| 10mM/100μL | In stock |
€650 |
Remdesivir triphosphate triethylammonium salt
Characterisation
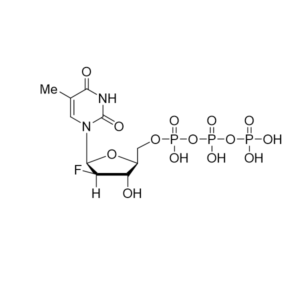
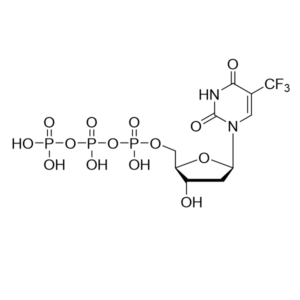
CAS: 2725956-16-9
IUPAC Name: 2-C-(4-Aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-2,5-anhydro-D-altrononitrile 6-triphosphate trisodium
Other names: GS-441524 triphosphate, Remdesivir metabolite, Remdesivir triphosphate metabolite, Remdesivir triphosphate triethylammonium salt
Molecular weight: 834.78 g/mol
Molecular formula: C30H61N8O13P3
Storage: -20 °C, protect from light, stored under argon
All our products are lyophilized to ensure long shelf life and the best shipping conditions. We already have tens of satisfied clients across three continents.
We are also providing Remdesivir triphosphate in the form of Na+ salt.
Description
Remdesivir triphosphate triethylammonium salt is synthesised by Santiago Lab (Prague, Czech Republic). GS-443902 is the active triphosphate metabolite of Remdesivir with activity against zoonotic feline infectious peritonitis virus (FIPV) and severe acute respiratory syndrome (SARS) virus from the Coronaviridae family. GS-443902 (GS-441524 triphosphate) is a potent viral RNA-dependent RNA-polymerases (RdRp) inhibitor with IC50s of 1.1 µM, 5 µM for RSV RdRp and HCV RdRp, respectively.
Remdesivir, developed by Gilead Sciences, is an adenosine prodrug that metabolizes into its active form GS-441524, which interferes with the action of viral RNA-dependent RNA polymerase and evades proofreading by viral exoribonuclease (ExoN), causing a decrease in viral RNA production.
Chemicals are distributed worldwide
Buy Remdesivir triphosphate triethylammonium salt now, get your order in 48 hours
- Shipping through DHL in 48 hours. We can also arrange shipping on dry ice.
- All compounds are safely and rigorously packed
- We are sending triphosphates every Monday
- Delivery time 24 hours in Europe, 24-48 hours to USA, Canada, Asia
Payment
- We are sending the invoice the same day as the shipment
- We can modify the invoice for the academic institution so that the order can be paid from grants
- Payment terms 30 days net
| References | References |
|---|
Related products:
-
Emtricitabine triphosphate – CAS 145819-92-7
Price range: €700 through €2500 -
Remdesivir triphosphate trisodium – GS-443902 – CAS 1355050-21-3
Price range: €650 through €5090 -
Remdesivir – CAS 1809249-37-3
Price range: €200 through €5900 -
Lamivudine triphosphate – CAS 143188-53-8
Price range: €450 through €3500 -
Aviromix
€700 -
Remdesivir triphosphate – GS-443902 – CAS 1355149-45-9
Price range: €650 through €5090 -
Remdesivir monophosphate – CAS 1911578-74-9
Price range: €1000 through €1500 -
Clevudine triphosphate – CAS 174625-00-4
Price range: €800 through €6500 -
Islatravir triphosphate – CAS 950913-56-1
Price range: €2500 through €6000 -
Remdesivir diphosphate – CAS 1911578-77-2
Price range: €2500 through €3500 -
Telbivudine triphosphate – CAS 152502-95-9
Price range: €800 through €6500 -
Trifluridine triphosphate – CAS 345-03-9
Price range: €800 through €6500