- No products in the cart.
DMTMM in amide bond formation
Amide bond formation is one the most widely occurring reactions in organic chemistry as well as in drug development. There are numerous ways to synthesize amides. However, their general principle is mostly the same – the reaction of the corresponding amine with carboxylic acid or its derivative. To perform this reaction selectively under mild conditions directly from carboxylic acid, compatible with highly derivatized substrates, the acid must first be activated (for example with DMTMM).

Ways of carboxylic acid activation
There are generally three ways of acid activation – via acyl chloride, mixed anhydride or activated ester (Fig. 1). The last of the mentioned methods is often performed chemoselectively under mild conditions using one of the readily available activating reagents. There are plenty of the reagents to choose from, and the most favoured ones generally belong into one of the following groups: carbodiimides (DCC, EDC), phosphonium salts (PyBOP), uronium and guanidinium salts (TBTU, HATU, TOTU), or triazine derivatives (cyanuric chloride, DMTMM) (Fig. 2).
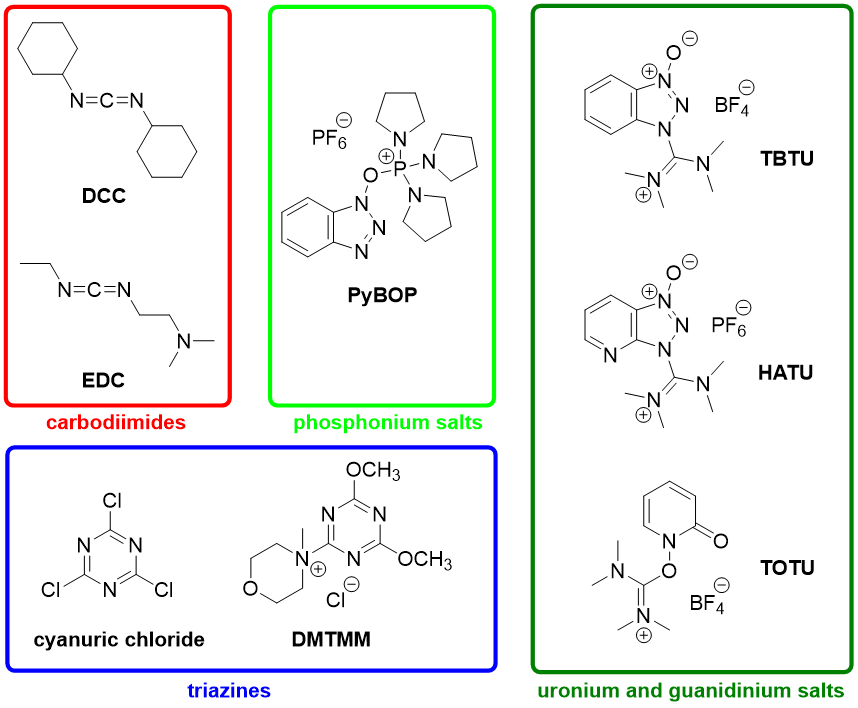
Each of these reagents has its advantages and disadvantages with respect to effectivity, toxicity, handling, racemization of substrates or by-product formation and product purification.
DMTMM – mechanism of activation
In this article, we would like to focus on triazine reagents, as they are often not the first choice for amide coupling reactions. Still, they also show interesting reactivity and properties. @Santiago lab, we have a very good experience with DMTMM, which we have repeatedly used in our latest project.
DMTMM (4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride, CAS: 3945-69-5) is air and water-stable solid that is usually used in the chloride form, but the tetrafluoroborate salt is also commercially available.
The significant advantage of DMTMM is the ability to promote amide bond formation selectively in protic solvents (water, alcohols). N-methylmorpholine (NMM) hydrochloride and 1-hydroxy-3,5-dimethoxytriazine are formed as by-products during the reaction. Both are water-soluble and easy to remove (Fig. 3).
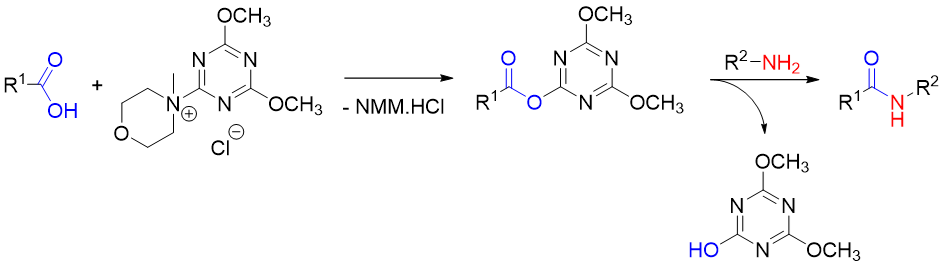
DMTMM – applications
In some cases, DMTMM has been shown to be preferentially over other coupling reagents, especially for the reactions of polysaccharides such as hyaluronic acid and for the coupling of sterically hindered amines. Moreover, it has also proved effective at reducing epimerization if chiral reagents are used. In addition to its use in amide formations, DMTMM can also be successfully employed to prepare other carboxylic acid derivatives such as esters and anhydrides.
DMTMM – preparation
DMTMM can also be generated in situ in the reaction mixture from its precursor CDMT (2-chloro-4,6-dimethoxy-1,3,5-triazine) and NMM. CDMT is also a stable, crystalline solid that is commercially available. It can be used as an activating reagent similar to DMTMM. It usually requires the use of a tertiary amine base for acid activation.
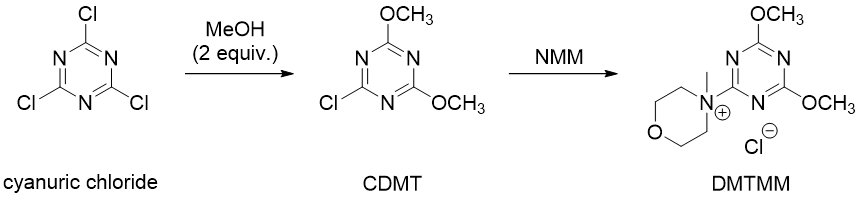
The simplest triazine activating reagent is cyanuric chloride (1,3,5-trichlorotriazine), which also serve as a starting derivative for the synthesis of CDMT and DMTMM (Fig. 4). It is one of the most cost-effective reagents for amide coupling, as it is produced on a large industrial scale. Moreover, it contains three reactive chlorine atoms in its structure, which can be employed in sub-stoichiometric quantities for acid activation. Similarly to DMTMM and CDMT, cyanuric chloride also tolerates the presence of water and the by-product cyanuric acid can be removed by an aqueous workup.
Santiago Lab
At Santiago Lab we are working on various interesting projects for broad range of the international clients where we employed DMTMM many times.
If you would like to know more, or we can help you with your research, write an email to Krystof Sigut on krystof.sigut@santiago-lab.com or reach him on the phone +420 776 750 591.
Drop us a line
We will respond as soon as possible, typically within 48 hours.
References
Amide coupling reviews:
Han, S.-Y.; Kim, Y.-A. Tetrahedron 2004, 60 (11), 2447–2467. http://dx.doi.org/10.1016/j.tet.2004.01.020
Montalbetti, C. A. G. N.; Falque, V. Tetrahedron 2005, 61 (46), 10827–10852. http://doi.org/10.1016/j.tet.2005.08.031
Valeur, E.; Bradley, M. Chem. Soc. Rev. 2009, 38 (2), 606–631. https://doi.org/10.1039/B701677H
El-Faham, A.; Albericio, F. Chem. Rev. 2011, 111 (11), 6557–6602. https://doi.org/10.1021/cr100048w
Lanigan, R. M.; Sheppard, T. D. Eur. J. Org. Chem. 2013, 2013 (33), 7453–7465. https://doi.org/10.1002/ejoc.201300573
Taylor, J. E.; Bull, S. D. In Comprehensive Organic Synthesis, 2nd ed.; Knochel, P., Molander, G. A. Eds.; Elsevier: New York, 2014; pp 427−478. https://doi.org/10.1016/B978-0-08-097742-3.00617-0
Dunetz, J. R.; Magano, J.; Weisenburger, G. A. Org. Process Res. Dev. 2016, 20 (2), 140−177. https://doi.org/10.1021/op500305s


