- No products in the cart.
Lawesson’s reagent
Many biologically active substances contain a sulphur atom; however, it is usually not so easy to introduce sulphur into the molecule. Therefore, several thionation reagents were developed in the past. The previously used phosphorus sulphide was successively substituted with 2,4-bis(4-methoxyphenyl)-1,3,2,4-dithiadiphosphetane-2,4-dithione (known these days as Lawesson’s reagent, Figure 1), which is used as a thionation reagent for transformations of carbonyl groups into the corresponding thiocarbonyl groups.
This compound was first prepared in 1956 (https://doi.org/10.1021/ja01600a058) and used as a thionation agent in 1967 (https://doi.org/10.1016/S0040-4039(00)90895-5). As a first reaction, the 2,4-bis(4-methoxyphenyl)-1,3,2,4-dithiadiphosphetane-2,4-dithione (Lawesson’s reagent) was successfully utilized for the transformation of benzophenone to thiobenzophenone.
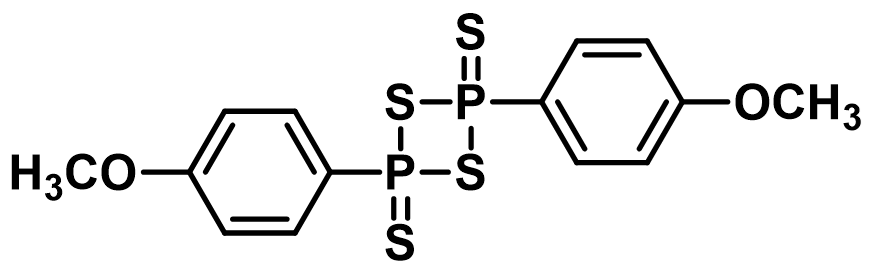
In 1978, Lawesson published a paper dealing with the synthesis of thioketone from ketones, which greatly expanded the use of this substance in organic synthesis. (https://doi.org/10.1002/bscb.19780870310). As a result, 2,4-bis(4-methoxyphenyl)-1,3,2,4-dithiadiphosphate-2,4-dithionate was named Lawesson’s reagent.
Preparation of Lawesson’s reagent
Lawesson’s reagent is prepared from anisole and phosphorus sulphide (Org. Synth. 1984, 62, 158; DOI: 10.15227/orgsyn.062.0158). Other possible preparation is from anisole, red phosphorus and sulphur (Mazitova, F. N.; Khairullin, V. K. Zh. Org. Khim. 1981, 51, 958.).
Lawesson’s reagent- Reaction mechanism
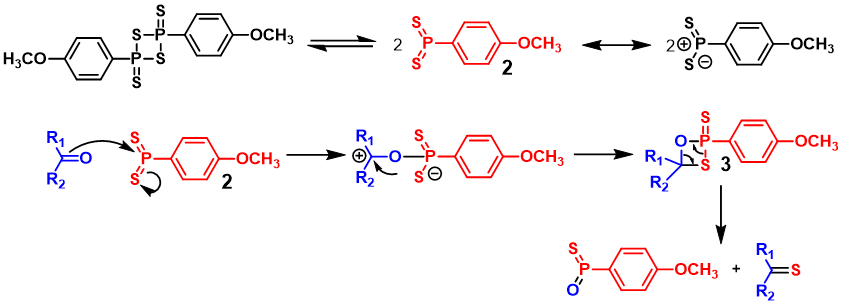
Lawesson’s reagent is in equilibrium with reactive intermediate 2 (dithiophosphine ylide). This ylide then reacts with the carbonyl group to form thiaoxaphosphetane intermediate 3, which is further decomposed by a cycloelimination mechanism as a Wittig-analogous reaction. (https://www.organic-chemistry.org/namedreactions/lawessons-reagent.shtm)
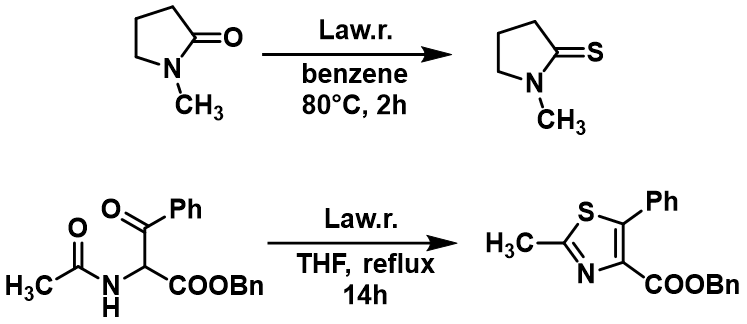
Lawesson’s reagent can convert an oxygen atom to a sulphur atom in a ketone, aldehyde, ester, amide, lactones, lactams, and quinones. Ketones, amides, and lactams react faster than corresponding esters (Figure 3). Typical conditions are using 2-3 equivalents of Lawesson’s reagent in a nonpolar solvent (toluene, xylene) heated to 80-100 °C. One of many examples of the use of Lawesson’s reagent is the thionation of N-methylpyrrolidone or cyclization reactions (Figure 3). (https://www.thieme-connect.de/products/ejournals/abstract/10.1055/s-2003-41447; https://pubs.acs.org/doi/10.1021/jo9016265).
Woollins’ reagent
Similar to Lawesson’s reagent, Wollins’ reagent (2,4-diphenyl-1,3,2,4-diselenadiphosphetane 2,4-diselenide (Figure 4) is used for the transformation of the oxygen atom of the carbonyl group to selenium. This reagent is named after John Derek Woollins (https://doi.org/10.1002/chem.200500291).
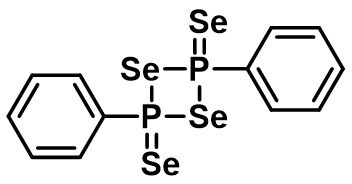
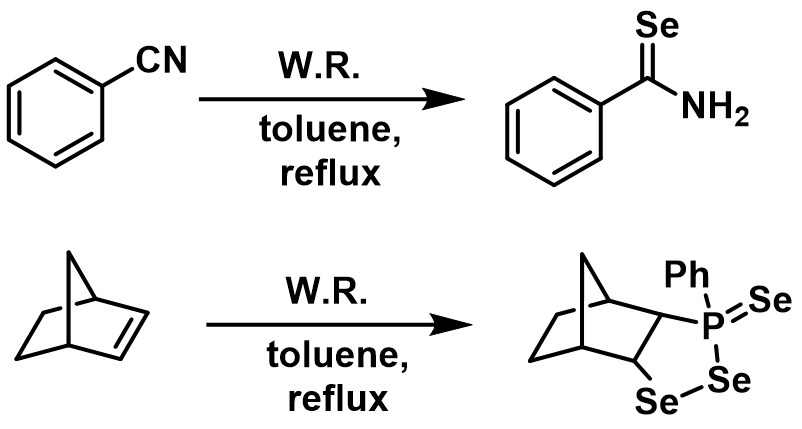
Woollins’ reagent is prepared by heating dichlorophenylphosphine and sodium selenide or by the reaction of the (PhP)5 with elemental selenium in toluene. Woollins’ reagent is used to convert amides, acids and ketones to the corresponding selenium derivatives. In addition, Woollins’ reagent also reacts with nitriles, alkenes and alkynes (Figure 5). (https://doi.org/10.1016/S0040-4039(01)01113-3; https://doi.org/10.1002/anie.200800572)
If you want to discover more about our services, discuss specific research (either with or without Lawesson’s or Woollins’ reagent, custom synthesis project or receive a proposal, please do not hesitate to write an email to Krystof Sigut at krystof.sigut@santiago-lab.com or reach him on the phone +420 776 750 591.
Drop us a line
We will respond as soon as possible, typically within 48 hours.


