- No products in the cart.
Reagent of the month – March – Seyferth-Gilbert and Bestmann-Ohira reagents
Dimethyl(1-diazomethyl)phosphonate (Seyferth–Gilbert reagent, Figure 1) is a homologation reagent used to convert aryl ketones and some aldehydes into alkynes elongated by one carbon atom coming from the reagent itself in the Seyferth–Gilbert homologation (Figure 2).
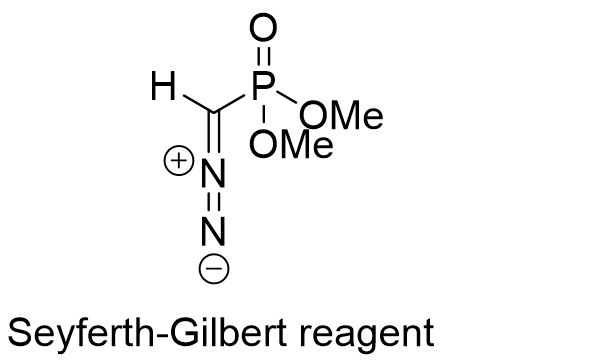
Reaction mechanism
First, the acidic proton of the Seyferth–Gilbert reagent (1) is deprotonated with a strong base (potassium tert-butoxide is usually used) giving the carbanion 2. Then, a mechanism similar to that of Horner–Wadsworth–Emmons reaction proceeds, leading to the formation of the corresponding diazoalkane 5. This intermediate then decomposes upon warming the reaction mixture giving the carbene intermediate 7 with a loss of nitrogen. Finally, the carbene undergoes a rearrangement giving the final alkyne 8. (Original publication: 10.1021/jo00349a007)
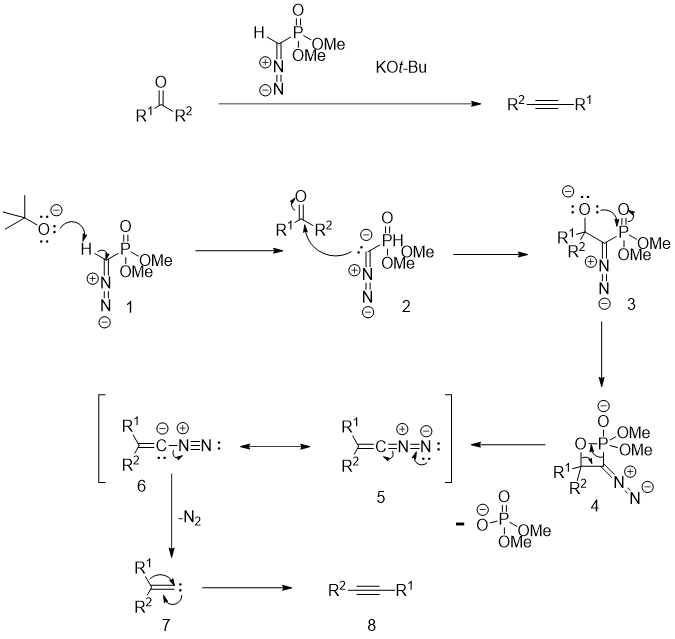
Limitations and updates
Due to the use of a strong base in the initial step, Seyferth–Gilbert homologation cannot be performed with base labile carbonyl compounds such as enolizable aldehydes, which would undergo aldol condensation reactions under basic conditions.
This problem has been overcome by a different way of generating the carbanion 2 in the reaction mixture. Methanolysis of dimethyl diazo-2-oxopropylphosphonate was first used by Susumu Ohira from the Okayama university to perform the analogous homologation reaction using a weaker base (https://doi.org/10.1080/00397918908050700).
Later, the reaction conditions were developed further by Bestmann et al. to run using just potassium carbonate as a base, making the reaction usable also for the synthesis of terminal alkynes from enolizable aldehydes (DOI: 10.1055/s-1996-5474, doi:10.1055/s-2003-44346). Dimethyl diazo-2-oxopropylphosphonate (Figure 3.) is called Ohira–Bestmann reagent.
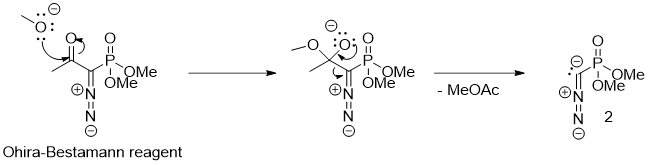
Preparation of the Ohira–Bestmann reagent
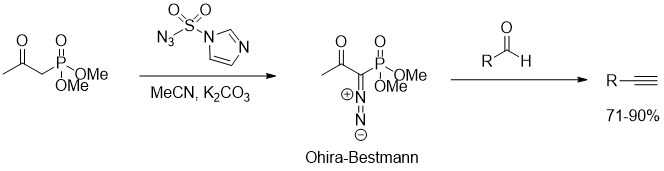
The Ohira–Bestmann reagent can be easily prepared in-situ by diazo transfer reagents such as sulfonyl azides (Figure 4., https://doi.org/10.1021/jo501803f)
Application in synthesis
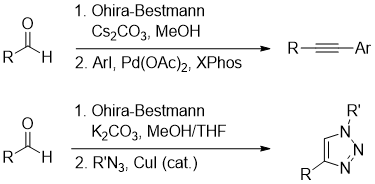
One pot tandem procedures combining Ohira–Bestmann homologation with reactions of terminal alkynes, such as sonogashira coupling or CuAAC reactions, were recently developed (Figure 5., https://doi.org/10.1021/acs.joc.9b01367, DOI: 10.1055/s-2007-990877).
We have a lot of experience at Santiago lab with various chemistry fields and specialized reagents. If you want to learn more about our services, discuss specific research or custom synthesis project, or receive a proposal, please contact Krystof Sigut at krystof.sigut@santiago-lab.com
or reach him on the phone +420 776 750 591.


