- No products in the cart.
Nucleoside triphosphates with antiviral activity
Viruses are causing many troubles in our lives these days. They cause many serious diseases (some of them even deadly) such as cold, influenza, EBOLA, AIDS or COVID-19.
At Santiago Lab, we are constantly trying to work on a synthesis of new compounds that might exhibit antiviral activity. Last year, the Central Bohemian Innovation Centre funded one of our projects, where we chose six known nucleosides with reported antiviral activity and transformed them to their corresponding biologically active triphosphates.
Even though these nucleosides have been known in the literature for some time, their triphosphates are only very limitedly commercially available or not available at all. Therefore, we developed unique synthetic pathways leading to these triphosphates, and now we would like to offer them for researchers worldwide dealing with antiviral research.
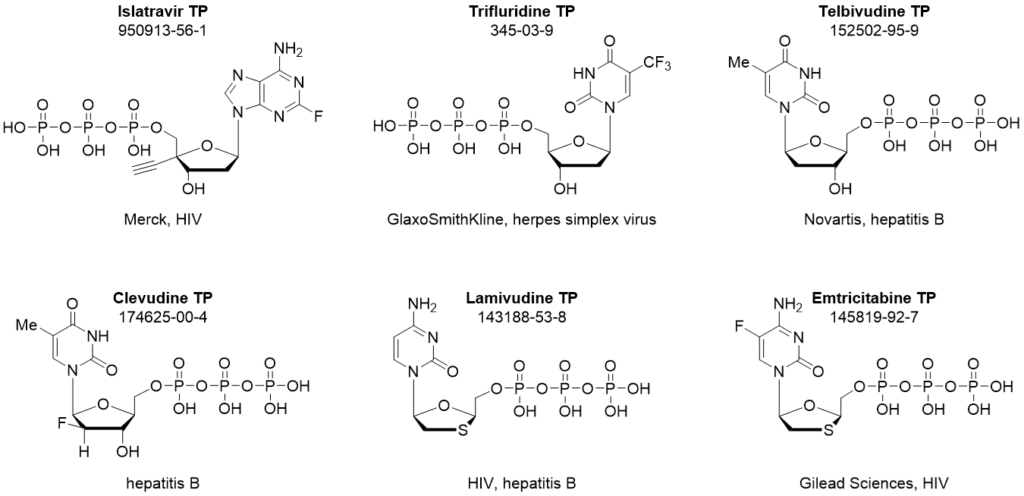
Islatravir triphosphate
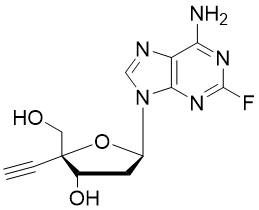
Islatravir (ISL, MK-8591, EFdA, CAS: 865363-93-5) is an investigational antiviral drug developed by Merck for the treatment of HIV. Currently, it is one of the hottest antiretroviral agents, as it has been advanced to phase 2 or 3 in several clinical trials for HIV treatment.
Islatravir triphosphate is in stock here.
Trifluridine triphosphate
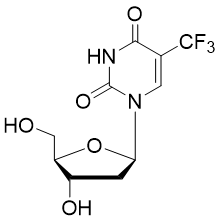
Trifluridine (also called trifluorothymidine; abbreviation TFT or FTD )is a fluorinated pyrimidine nucleoside structurally related to idoxuridine. It is an active antiviral agent in ophthalmic solutions used mainly to treat primary keratoconjunctivitis and recurrent epithelial keratitis due to the herpes simplex virus. It displays effective antiviral activity against Herpes simplex virus type 1 and 2.
In anticancer therapy, trifluridine acts as a thymidine-based nucleoside metabolic inhibitor that gets incorporated into DNA of cancer cells following cell uptake to aberrate DNA function during cell replication. It is also a component of the anti-cancer drug trifluridine/tipiracil, which is taken by mouth.
Trifluridine triphosphate is in stock here.
Telbivudine triphosphate
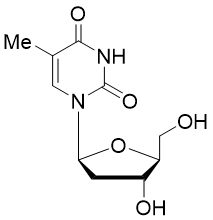
Telbivudine is a synthetic thymidine β-l-nucleoside analogue; it is the l-isomer of thymidine. Telbivudine impairs hepatitis B virus (HBV) DNA replication by leading to chain termination. It differs from the natural nucleotide only with respect to the location of the sugar and base moieties, taking on a levorotatory configuration versus a dextrorotatory configuration, as do the natural deoxynucleosides.
Telbivudine triphosphate is in stock here.
Clevudine triphosphate
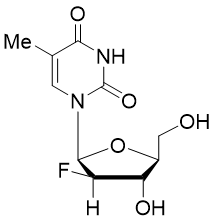
Clevudine (also known as Levovir and Revovir) is a synthetic pyrimidine analogue with activity against the hepatitis B virus (HBV). The triphosphate metabolite competes with thymidine for incorporation into viral DNA, causing DNA chain termination and inhibiting the function of the hepatitis B virus (HBV) DNA polymerase (reverse transcriptase). Clevudine is also studied to evaluate the efficacy and safety against a moderate form of COVID-19.
Clevudine triphosphate is in stock here.
Lamivudine triphosphate
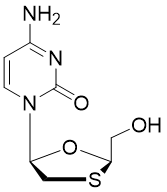
Lamivudine, commonly called 3TC, a reverse transcriptase inhibitor and zalcitabine analogue in which a sulphur atom replaces the 3’ carbon of the pentose ring. It is an antiretroviral medication used to prevent and treat HIV/AIDS (HIV-1 and HIV-2). It is also used to treat chronic hepatitis B when other options are not possible. Lamivudine is typically used in combination with other antiretrovirals such as zidovudine and abacavir.
Lamivudine was patented in 1995 and approved for use in the United States in 1995. It is on the World Health Organization’s List of Essential Medicines. It is available as a generic medication.
Lamivudine triphosphate is in stock here.
Emtricitabine triphosphate
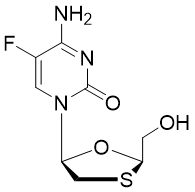
Emtricitabine (commonly called FTC, trade name Emtriva) is a cytidine analogue that acts as a nucleoside reverse-transcriptase inhibitor (NRTI) for the prevention and treatment of HIV infection in adults and children.
It is also marketed in a fixed-dose combination with tenofovir disoproxil (Viread) under the brand name Truvada, and with tenofovir alafenamide (Vemlidy) under the brand name Descovy.
Emtricitabine triphosphate is in stock here.
Santiago Lab
All nucleoside triphosphates were prepared in excellent purity (C18 chromatography together with ion-exchange chromatography). Our triphosphates are lyophilized to ensure long shelf life and the best stability during shipment (usually on dry ice to most countries worldwide).
If you would like to know more, or we can help you with your research, write an email to Krystof Sigut on krystof.sigut@santiago-lab.com or reach him on the phone +420 776 750 591.
Drop us a line
We will respond as soon as possible, typically within 48 hours.
References
Markowitz M, Sarafianos SG. “EFdA (4′-ethynyl-2-fluoro-2′-deoxyadenosine, MK-8591): a novel HIV-1 reverse transcriptase translocation inhibitor”, Curr Opin HIV AIDS. 2018;13(4):294-299, doi:10.1097/COH.0000000000000467
Kawamoto, A; Kodama, E; Sarafianos, SG; Sakagami, Y; Kohgo, S; Kitano, K; Ashida, N; Iwai, Y; Hayakawa, H; Nakata, H; Mitsuya, H; Arnold, E; Matsuoka, M (2008). “2′-deoxy-4′-C-ethynyl-2-halo-adenosines active against drug-resistant human immunodeficiency virus type 1 variants”. The International Journal of Biochemistry & Cell Biology. 40 (11): 2410–20, doi:10.1016/j.biocel.2008.04.007.
World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization.
Anuj K. Patel, Ritika Abhyankar, Lauren K. Brais, Mei Sheng Duh, Victoria E. Barghout, Lynn Huynh, Mihran A. Yenikomshian, Kimmie Ng, Charles S. Fuchs, “Trifluridine/Tipiracil and Regorafenib in Patients with Metastatic Colorectal Cancer: A Retrospective Study at a Tertiary Oncology Center”, The Oncologist, Volume 26, Issue 12, December 2021, 2161–2169, https://doi.org/10.1002/onco.13942.
Henry L.Y. Chan, E. Jenny Heathcote, Patrick Marcellin, et al. “Treatment of Hepatitis B e Antigen–Positive Chronic Hepatitis with Telbivudine or Adefovir: A Randomized Trial”, Ann Intern Med. 2007;147:745-754, doi:10.7326/0003-4819-147-11-200712040-00183

